.Yaohai Bio-Pharma has established robust and mature platforms for plasmid production, mRNA synthesis, and LNP formulation. The company provides end-to-end solutions to global clients across key development stages, including early R&D, clinical-stage process development, GMP manufacturing, analytical testing, drug product fill-finish, and regulatory submission support.
CRDMO Modalities
Microbial CRDMO Solutions with excellence in quality and global regulatory compliance
mRNA
Yaohai Bio-Pharma established its mRNA CRO platform in 2022 and has supported over 800 RNA projects to date, partnering with universities, research institutes, and many biotech companies. Leveraging GMP plasmid production, robust IVT systems, advanced purification, and LNP delivery technologies, we now provide integrated mRNA solutions from early R&D to commercial manufacturing, accelerating project timelines for global clients.
CRDMO Modalities
Service Platform
Service Highlights
CRDMO Modalities
Building on 10+ years of experience in mRNA field and fully integrated mRNA CRDMO capabilities, Yaohai Bio-Pharma proudly present the “RNASUP” CRDMO platform—covering plasmid preparation, mRNA synthesis & purification, LNP encapsulation, GMP production, and regulatory support—delivering seamless services for
- Protein Replacement Therapies
- mRNA-encoded Immunogens
- Antibody Therapeutics (e.g., T-cell Engagers)
- mRNA Vaccines
- In Vivo CAR Therapies
- In Vivo Gene Editing
Our end-to-end mRNA CRDMO platform supports the project from R&D (mg) to commercial (tens of grams) scale, fully meeting EMA and FDA GMP requirements.
“RNASUP” CRDMO platform
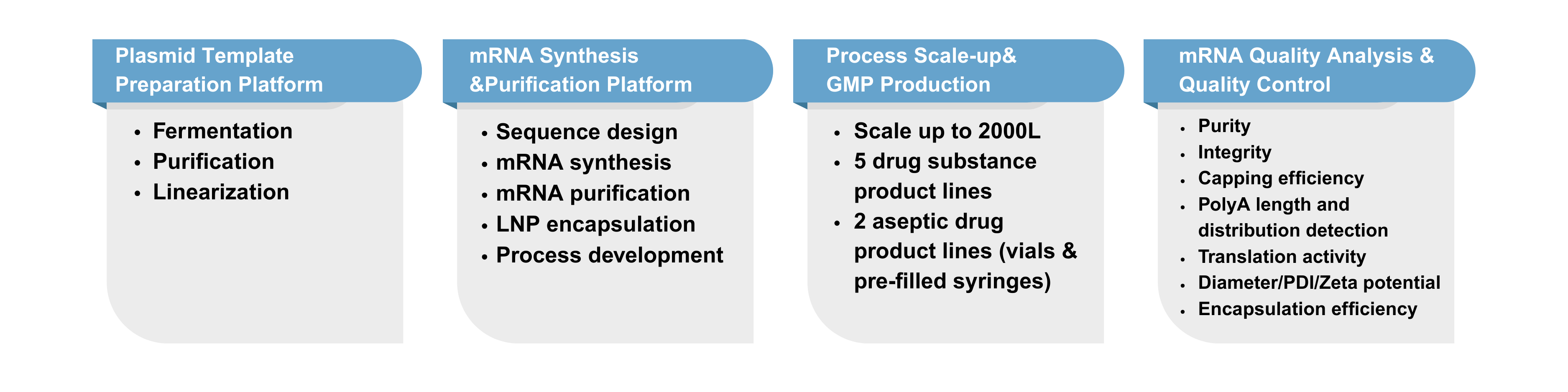
Service Platform
Microbial CRDMO Service Platform for mRNA
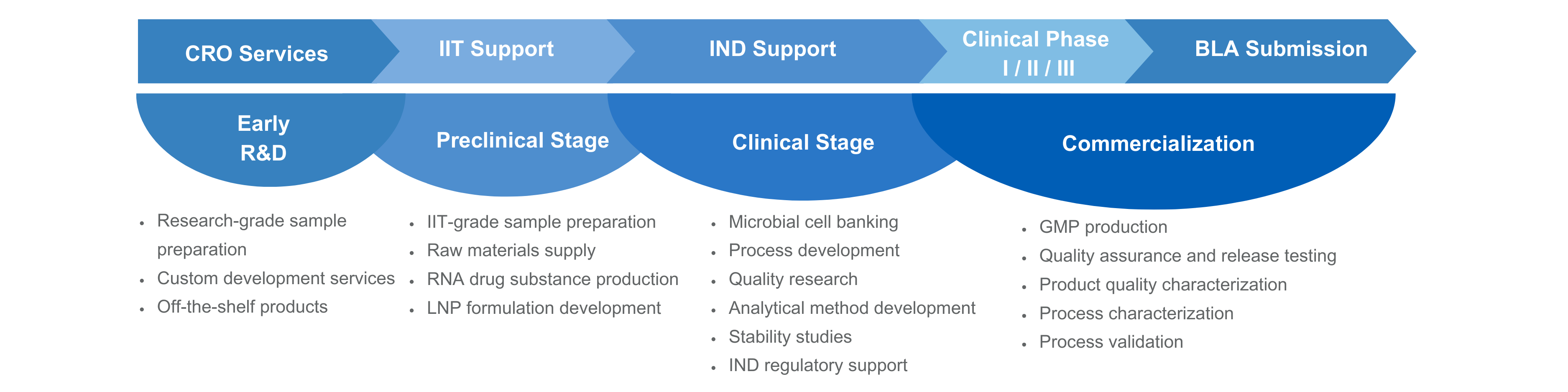
Service Highlights
End-to-End mRNA Solutions
- Our solutions span from plasmid, mRNA drug substance preparation, to LNP-formulated drug product production
- Seamless coordination across all stages ensures efficient and reliable project delivery
Comprehensive Lifecycle Support
- From process development and GMP production to quality studies and regulatory consulting, we provide support to accelerate your project
- Flexible CRDMO solutions tailored to project needs
Strict Quality System
- Leveraging the experience, Yaohai have established scalable, high-capping-efficiency, and high-integrity mRNA platform that minimizes dsRNA residuals and other impurities—enabling rapid progression from PCC to commercialization
Global Compliance
- cGMP facilities compliant with US and EU GMP standards, with multiple successful regulatory experiences
mRNA CRDMO Service Cases

Related products
Let's start talking with us
Our value
Serve With Heart
Build Future Together
Yaohai Bio-Pharma delivers tailored solutions for global clients, meeting diverse project requirements with excellence and efficiency.
Proven Project Excellence
Proven track record of over 100 successful projects, spanning preclinical studies and Phase I/II/III clinical trials, including US–China dual submissions and Australian regulatory filings.
Regulatory Compliance Assurance
A robust GMP-aligned quality system with standardized SOPs, fully compliant with global regulatory guidelines across the entire product lifecycle.
Expert Team Support
Powered by industry-leading scientists and cross-functional experts, our team delivers CRDMO projects with speed, precision, and deep technical insight.