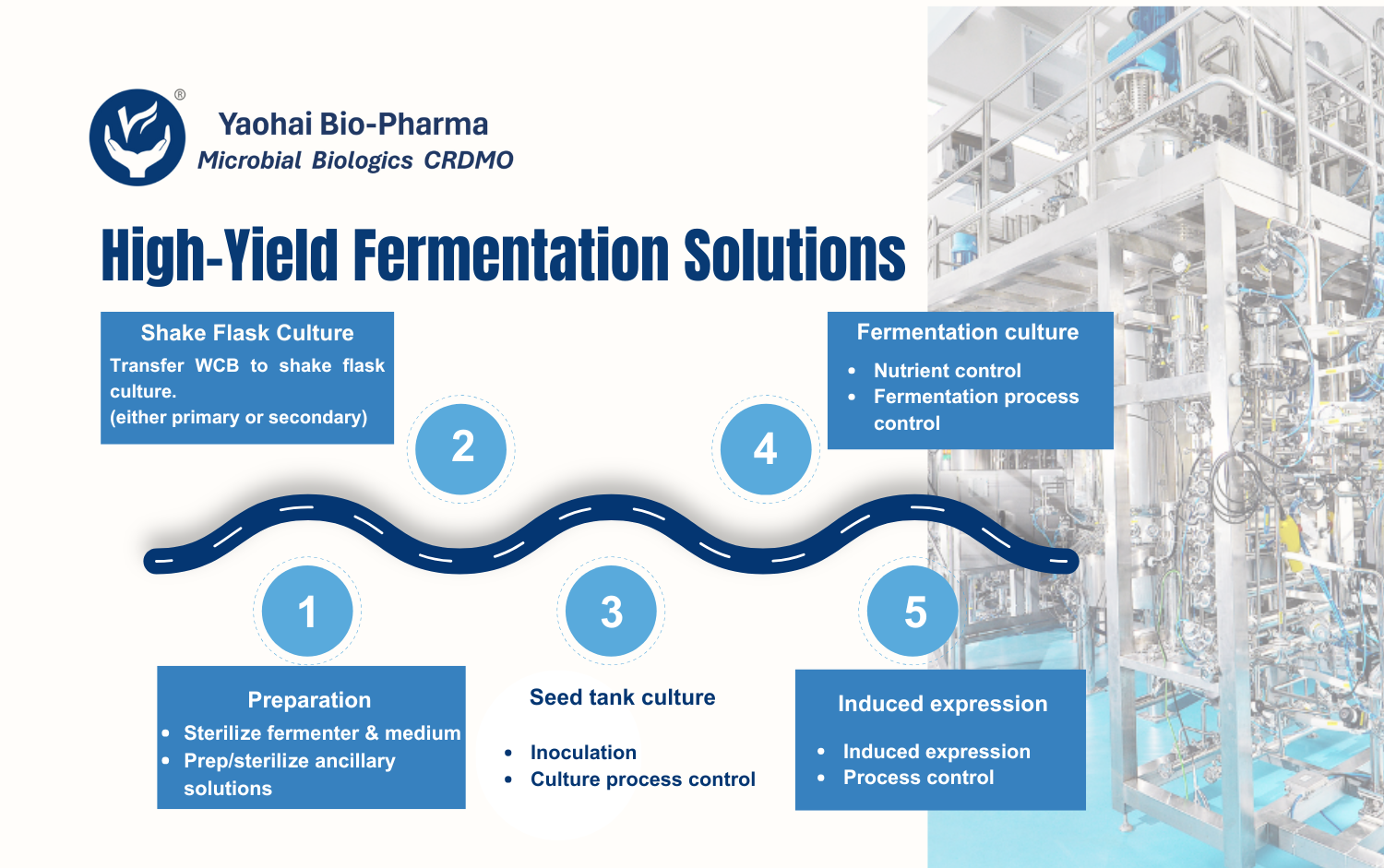
Yaohai Bio-Pharma is a leading CRDMO specializing in microbial expression systems. We offer end-to-end purification process development, starting from primary purification and moving all the way to the final drug substance. Our goal is simple: deliver robust, scalable, and compliant purification processes that meet each client’s needs.
Because we follow Quality by Design (QbD) principles, we use both OFAT and Design of Experiments (DoE) to evaluate key parameters. As a result, we can identify the best conditions faster and with greater accuracy. Whether the target protein is intracellular, extracellular, or found in inclusion bodies, our platform ensures high purity, strong biological activity, and consistent impurity removal.
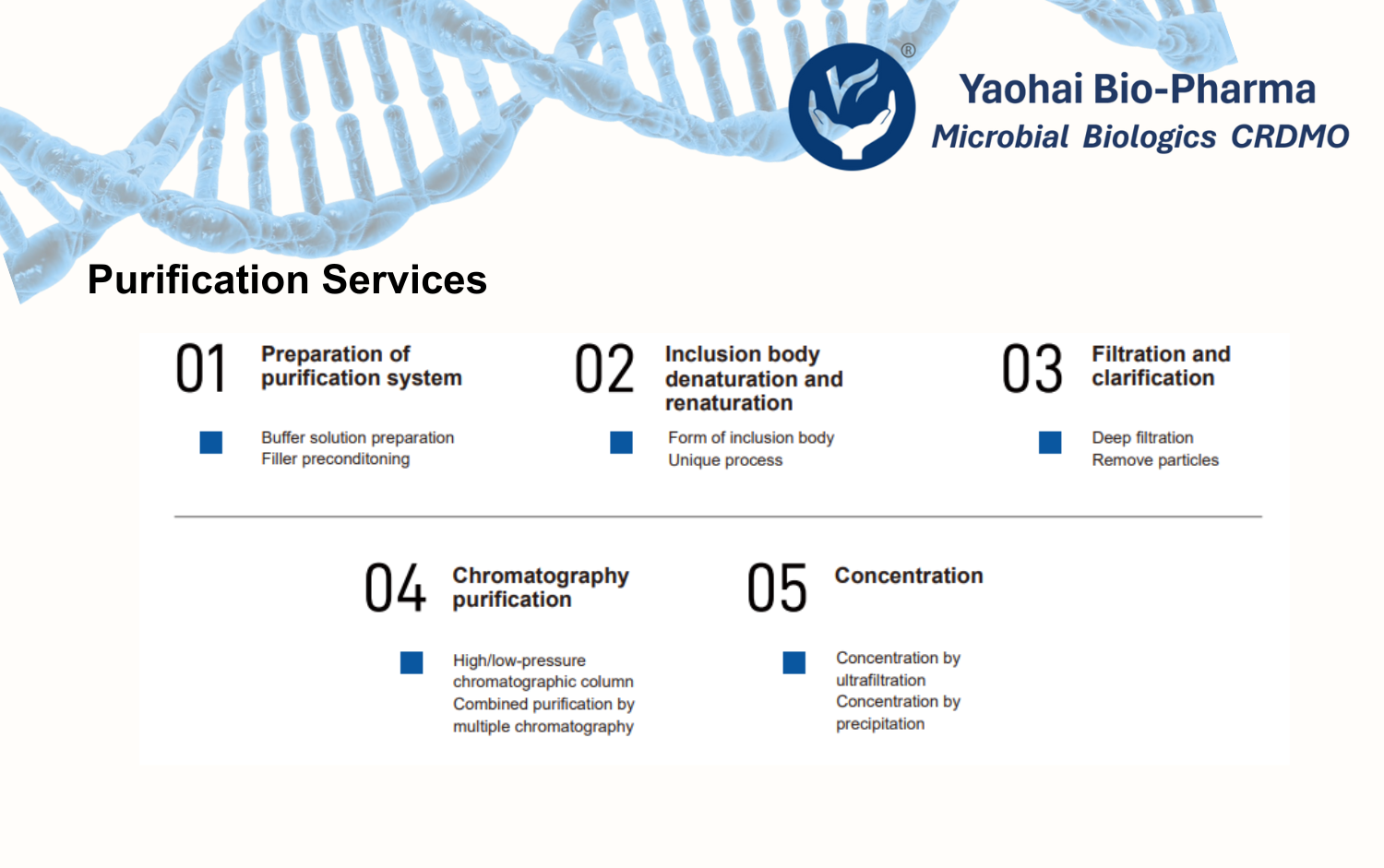
Comprehensive Purification Capabilities
Centrifugation and filtration
High-pressure homogenization
Protein precipitation
Inclusion body solubilization and refolding
Chromatography (IEX, HIC, affinity, mixed-mode)
Ultrafiltration and diafiltration
Together, these steps create a smooth, step-by-step transition from crude purification to final polishing. This approach helps us deliver high-quality recombinant protein bulk drug substances with reliability and reproducibility.
High-Throughput Resin Screening Platform
To speed up early development, Yaohai Bio-Pharma uses a 96-well high-throughput screening platform. This system tests large resin libraries—including anion exchange, cation exchange, hydrophobic interaction, and mixed-mode resins. As a result, resin selection becomes faster, clearer, and more efficient.
Rapid Chromatography Process Development
Our platform includes more than ten AKTA Avant and AKTA Pure systems. With these tools, we perform DoE-based or OFAT optimization. This helps us determine the best column sequence and operating parameters. Consequently, we can shorten development timelines and prepare processes for smooth tech transfer.
Extensive Experience in Complex Modalities
Our team has deep experience in chromatography optimization, inclusion body refolding, and VLP disassembly and reassembly. Therefore, we can support diverse and complex biologic modalities, such as vaccines, peptide hormones, cytokines, antibody fragments, and allergens.
Integrated CRDMO Support
With more than ten years of commercial manufacturing experience, Yaohai Bio-Pharma always designs purification processes with scale-up in mind. In most cases, our small-scale process includes no more than four chromatography steps. This approach ensures a stable and predictable transition from development to pilot-scale and GMP manufacturing.
About Yaohai Bio-Pharma
Yaohai Bio-Pharma is a CRDMO with extensive expertise in microbial expression systems. We focus on recombinant proteins, nucleic acid drugs, nanobodies, plasmid DNA, and innovative vaccines. From R&D to commercial manufacturing, we offer full-lifecycle solutions. Our mission is to build an open, integrated CRO/CDMO/MAH platform that supports global partners from discovery to market.